Europe Nimbin Market Size, Share, Trends & Growth Forecast Report Segmented By Application (Pharmaceutical & Nutraceutical Segment, Cosmetics & Personal Care Segment), Source, And Country (UK, France, Spain, Germany, Italy, Russia, Sweden, Denmark, Switzerland, Netherlands, Turkey, Czech Republic & Rest Of Europe), Industry Analysis From 2026 To 2034
Europe Nimbin Market Size
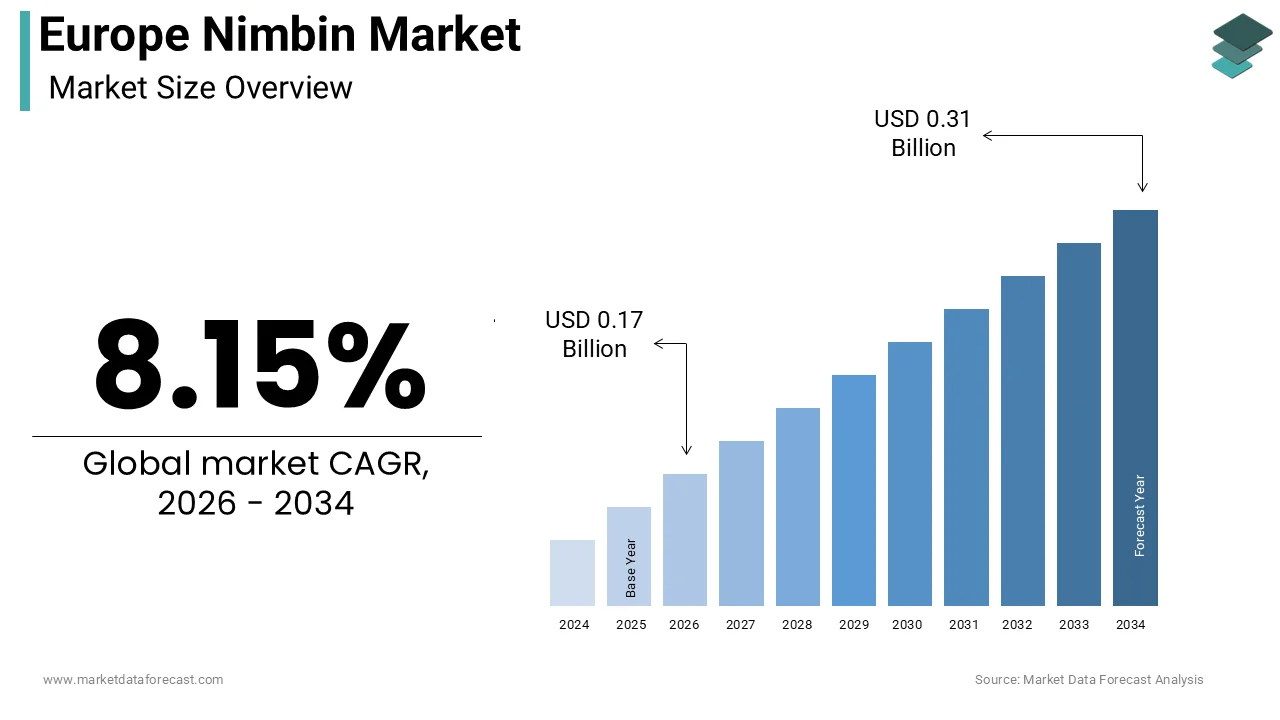
The Europe Nimbin market size was calculated to be USD 0.15 billion in 2025 and is anticipated to be worth USD 0.31 billion by 2034, from USD 0.17 billion in 2026, growing at a CAGR of 8.15% during the forecast period.
Nimbin is a pivotal triterpenoid compound derived principally from the seeds and leaves of the neem tree, scientifically classified as Azadirachta indica. This bioactive molecule demonstrates robust anti-inflammatory, antiviral, and antifungal characteristics that have attracted substantial interest within the European pharmaceutical and cosmetic industries. The extraction, purification, and application of this specific compound in high-value sectors, including dermatology, agrochemicals, and personal care. According to the European Medicines Agency, traditional herbal medicinal products containing neem derivatives are officially recognized for their beneficial effects on skin health. The agricultural cultivation of neem is not indigenous to Europe, which means the supply chain depends heavily on imports from Asia and Africa. As per the Food and Agriculture Organization of the United Nations, global neem production surpasses 1 million tons annually, with India standing as the largest producer. This dependency establishes a unique dynamic in the European market where quality assurance and supply chain transparency are critical. The incorporation of nimbin into advanced drug delivery systems and organic skincare formulations marks the current technological frontier.
MARKET DRIVERS
Rising Prevalence of Skin Disorders Drives Demand for Natural Therapeutics
The increasing incidence of dermatological conditions for the adoption of nimbin-based treatments is the primary factor accelerating the growth of Europe nimbin market. Chronic skin ailments, such as eczema, psoriasis, and acne, affect a significant segment of the European population, creating a sustained need for effective therapeutic solutions. According to the International Federation of Psoriasis Associations, approximately 2% of the European population suffers from psoriasis, which equates to nearly 15 million individuals. Conventional synthetic treatments often involve adverse side effects, leading patients and healthcare providers to seek safer natural alternatives. Nimbin exhibits strong anti-inflammatory properties that assist in reducing redness, swelling, and irritation linked to these conditions. The European Society for Dermatology and Psychiatry indicates that stress induced skin disorders have risen by 20% in the last decade, further intensifying the need for calming agents. Consumers are becoming increasingly aware of the advantages of neem-derived compounds due to extensive educational initiatives by dermatologists and wellness influencers. This awareness stimulates the formulation of specialized skincare products containing standardized nimbin extracts. Pharmaceutical firms are investing in clinical trials to confirm the efficacy of nimbin in treating resistant skin infections. The movement towards personalized medicine also supports this trend, as patients prefer tailored natural remedies over broad-spectrum antibiotics.
Expansion of the Organic Cosmetics Sector Fuels Ingredient Innovation
The vigorous growth of the organic and natural cosmetics industry, as a crucial active ingredient, is propelling the growth of Europe nimbin market. European consumers are showing a marked preference for clean-label products that exclude synthetic chemicals, parabens, and sulfates. Nimbin aligns perfectly with this narrative due to its natural origin and multifunctional benefits, including antimicrobial and antioxidant activities. Cosmetic formulators are utilizing nimbin to develop premium anti-aging and acne control products that appeal to environmentally conscious buyers. The European Commission’s regulations on cosmetic products require strict safety assessments, which nimbin meets due to its long history of safe use in traditional medicine. Major beauty brands are reformulating their product lines to include botanical extracts that offer proven efficacy. This consumer behavior encourages manufacturers to source high-quality nimbin extracts that satisfy rigorous purity standards. Innovations in extraction technologies, such as supercritical fluid extraction, enable the retention of nimbin’s bioactivity without using harmful solvents. This technological progress supports the creation of high-performance organic products. The alignment of Nimbin with sustainability goals further enhances its attractiveness to brands aiming to lower their environmental footprint.
MARKET RESTRAINTS
Supply Chain Vulnerabilities Due to Geographic Dependency
The heavy dependence on imported raw materials from non-European regions is restricting the growth of Europe nimbin market. Neem trees do not flourish in the temperate climate of Europe, necessitating the importation of neem seeds and leaves mainly from India, Nigeria, and other tropical countries. This geographic reliance exposes the market to various supply chain risks, including logistical disruptions, political instability, and trade barriers. Any fluctuation in the agricultural output of source countries due to climate change or pest infestations directly affects the consistency of nimbin supply. For example, droughts in key producing regions can cause a 30% reduction in neem seed yield, as reported by the recent survey. These variations lead to price volatility, which complicates budgeting and pricing strategies for European manufacturers. Additionally, the long transportation routes increase the carbon footprint of the final product, contradicting the sustainability claims often associated with natural ingredients. Regulatory hurdles at customs can further delay shipments, resulting in production halts. The absence of local cultivation options restricts the ability of European companies to secure a stable and controllable supply base. Companies must invest in complex inventory management systems to mitigate these risks, which raises operational costs.
Stringent Regulatory Compliance and Standardization Hurdles
The complex regulatory framework governing novel food ingredients and cosmetic substances is also hampering the growth of Europe nimbin market. Although neem has a history of traditional use, obtaining approval for specific purified compounds like nimbin requires extensive documentation and safety data. The European Food Safety Authority and the European Chemicals Agency enforce rigorous standards that can be time-consuming and expensive to meet. This barrier to entry discourages smaller players from innovating with nimbin-based formulations. Furthermore, there is a lack of standardized testing methods for quantifying nimbin content in various extracts, leading to inconsistencies in product quality. The absence of a unified European pharmacopoeia monograph for nimbin means that manufacturers must rely on internal standards, which may not be universally accepted. This ambiguity creates uncertainty for regulators and consumers alike. As per the European Directorate for the Quality of Medicines, harmonizing standards for botanical extracts remains an ongoing challenge. Non-compliance with these evolving regulations can result in product recalls and legal penalties, damaging brand reputation. The need for continuous monitoring of regulatory changes adds to the administrative burden on companies. These compliance requirements slow down the speed to market for new nimbin applications.
MARKET OPPORTUNITIES
Integration into Advanced Drug Delivery Systems
The integration into advanced drug delivery systems, such as nanocarriers and liposomes, is also expected to promote new opportunities for the growth of Europe nimbin market. These technologies enhance the bioavailability and targeted release of nimbin, improving its therapeutic efficacy in treating skin diseases and infections. According to the Journal of Controlled Release, nanoencapsulation can increase the stability of hydrophobic compounds like nimbin by 40%, making them more effective in topical applications. European research institutions are actively investigating these formulations to overcome the limitations of traditional extracts. The growing investment in biotechnology within the European Union supports this innovation. This funding facilitates collaborations between academic researchers and private companies to develop next-generation nimbin-based therapies. The ability to deliver nimbin directly to affected cells minimizes systemic side effects and enhances patient compliance. Pharmaceutical companies are seeking partnerships to license these advanced formulations for commercial use. The patent landscape for nimbin delivery systems is expanding, offering competitive advantages to early movers. This technological evolution opens new revenue streams beyond conventional cosmetics and supplements.
Growing Demand for Sustainable Agrochemical Solutions
The increasing adoption of biopesticides in agriculture is certainly contributing to fuel the growth of the Europe nimbin market. Nimbin is a key component in neem-based biopesticides, which provide an eco-friendly alternative to synthetic chemicals. The European Union’s Farm to Fork Strategy aims to reduce the use of chemical pesticides by 50% by 203,0 driving the demand for natural alternatives. Farmers are increasingly turning to neem-based products to manage pests while maintaining soil health and biodiversity. Nimbin acts as an antifeedant and growth regulator for insects, providing effective crop protection without harming beneficial organisms. This aligns with the principles of integrated pest management, which is widely promoted in Europe. Regulatory support for low-risk substances further accelerates the approval of nimbin-based agrochemicals. As per the European Food Safety Authority, several neem extracts have been approved for use in organic farming. This regulatory endorsement boosts consumer confidence and market acceptance. Agrochemical companies are expanding their portfolios to include nimbin-rich formulations catering to the organic farming sector. The shift towards sustainable agriculture practices ensures a steady demand for nimbin in the agritech segment.
MARKET CHALLENGES
Lack of Clinical Data for Specific Medical Claims
The scarcity of robust clinical data supporting specific medical claims is also one of the major challenges for the growth of Europe nimbin market. While traditional use suggests numerous health benefits, regulatory bodies require evidence from randomized controlled trials to approve therapeutic indications. According to the Cochrane Library, there are limited high-quality studies on the efficacy of purified nimbin in humans compared to crude neem extracts. This gap in scientific evidence restricts the ability of manufacturers to make definitive health claims on product labels. Without such claims, marketing efforts are limited to general wellness rather than targeted treatment, which reduces the perceived value of the product. Healthcare professionals are hesitant to recommend nimbin-based treatments without strong clinical backing. The cost of conducting large-scale clinical trials is prohibitive for many small and medium-sized enterprises operating in this space. As per the European Federation of Pharmaceutical Industries and Associations, the average cost of bringing a new drug to market exceeds 1 billion euros. Although nimbin is not a new chemical entity, proving its efficacy for specific indications requires significant investment. This financial barrier slows down the translation of traditional knowledge into modern medical applications. The lack of standardized clinical protocols for evaluating botanical compounds further complicates the research process. Until more comprehensive data is available, the market will struggle to gain full acceptance in the mainstream medical community.
Consumer Awareness Regarding Efficacy and Purity
The consumer awareness regarding the efficacy and purity of nimbin products is expected to further degrade the growth of Europe nimbin market. The varying qualities of neem extracts lead to confusion and mistrust among buyers. According to a survey, a UK consumer organization 60% of respondents expressed concern about the authenticity of natural ingredients in skincare products. Instances of adulteration or mislabeling have eroded consumer confidence in botanical supplements and cosmetics. Nimbin, being a specialized compound,d is often present in trace amounts in generic neem oils, leading to inconsistent results. Consumers who do not experience immediate benefits may dismiss the ingredient as ineffective. The lack of clear labeling standards for nimbin concentration exacerbates this issue. As per the European Consumer Organisation, inconsistent product quality is a top complaint in the natural cosmetics sector. Educating consumers about the difference between crude neem oil and standardized nimbin extracts requires substantial marketing expenditure. Brands must invest in third-party certifications and transparent sourcing practices to build trust. However, these measures increase the final product cost, potentially pricing out price-sensitive customers. The prevalence of counterfeit products in online marketplaces further complicates the landscape.
REPORT COVERAGE
| REPORT METRIC | DETAILS |
| Market Size Available | 2025 to 2034 |
| Base Year | 2025 |
| Forecast Period | 2026 to 2034 |
| CAGR | 8.15% |
| Segments Covered | By Application, Source, And Region |
| Various Analyses Covered | Global, Regional & Country Level Analysis; Segment-Level Analysis; DROC, PESTLE Analysis; Porter’s Five Forces Analysis; Competitive Landscape; Analyst Overview of Investment Opportunities |
| Regions Covered | UK, France, Spain, Germany, Italy, Russia, Sweden, Denmark, Switzerland, Netherlands, Turkey, and the Czech Republic |
| Market Leaders Profiled | EID Parry (India) Limited, Neem Biotech, Gramin India AgriBusiness, Ozone Biotech, P.J. Margo Pvt. Ltd., Fortune Biotech Ltd., Agro Extracts Limited, Nimtree Organics Pvt. Ltd., The Indian Neem Tree Company, Parker Biotech Private Limited |
SEGMENTAL ANALYSIS
By Application Insights
The pharmaceutical and nutraceutical segment was the largest by holding a prominent share of the Europe nimbin market in 2025 due to the increasing integration of botanical extracts into modern therapeutic formulations. Nimbin is extensively utilized for its anti-inflammatory and immunomodulatory properties, which are critical in managing chronic conditions such as arthritis and autoimmune disorders. According to the European Federation of Pharmaceutical Industries and Associations, the demand for plant-derived active pharmaceutical ingredients has grown by 8% annually over the past five years. Nutraceutical companies are incorporating standardized nimbin extracts into dietary supplements aimed at boosting immunity and reducing systemic inflammation. The regulatory approval of neem-based products for specific health claims further validates their use in this sector. Researchers are exploring the potential of nimbin in antiviral therapies, which has gained momentum following recent global health crises. The ability of nimbin to inhibit viral replication makes it a candidate for adjunctive treatments in respiratory infections. Pharmaceutical manufacturers are investing in purification technologies to achieve the high purity levels required for clinical applications.
The cosmetics and personal care segment is expected to register the fastest CAGR of 12.3% from 2026 to 2034, with a rapidly expanding avenue for nimbin utilization driven by the clean beauty movement and consumer preference for natural skincare solutions. Nimbin is valued for its antimicrobial and antioxidant properties, which make it an ideal ingredient for acne treatment, anti-aging creams, and soothing lotions. Consumers are increasingly seeking products that address skin concerns without relying on synthetic additives such as parabens and sulfates. Nimbin’s ability to regulate sebum production and reduce inflammation makes it particularly effective in formulations for oily and acne-prone skin. Major cosmetic brands are launching specialized lines featuring neem extracts to cater to this demand. As per Mintel, 40% of European consumers prefer skincare products with visible natural ingredients. The versatility of nimbin allows it to be incorporated into various product formats, including serum,s masks, and cleansers. Formulators are leveraging advanced extraction methods to ensure the stability and efficacy of nimbin in these products. The trend towards holistic wellness extends to skincare, where consumers view topical applications as part of overall health maintenance. This segment is characterized by high innovation rates and frequent product launches.
By Source Insights
Neem was accounted in holding a significant share of the Europe nimbin market in 2025. Seeds constitute the primary source for nimbin extraction due to their higher concentration of the compound compared to other parts of the tree. The oil extracted from neem seeds contains significant amounts of nimbin along with other limonoids, making it the preferred raw material for industrial applications. According to the Indian Council of Agricultural Research, neem seeds contain approximately 0.5 to 1% nimbin by weight, which is significantly higher than in leaves or bark. European manufacturers prioritize seed-derived extracts for their consistency and potency, which are crucial for meeting quality standards in pharmaceuticals and cosmetics. The supply chain for neem seeds is well established, with major imports coming from India and West Africa. This dominance ensures a steady flow of raw materials, although it also introduces dependency risks. Extraction processes for seed-derived nimbin are optimized to maximize yield and purity using techniques such as solvent extraction and chromatography. The resulting extracts are highly valued for their standardized nimbin content, which facilitates precise dosing in final products. Quality control measures are stringent to ensure the absence of contaminants such as aflatoxin,s which can occur in improperly stored seeds. The premium pricing of seed-derived nimbin reflects its superior quality and efficacy. This source segment dominates the market due to its proven track record and widespread acceptance in various industries. The
Neem leaves segment is also growing at an anticipated CAGR of 8.3% from 2026 to 2034. Despite this limitation, leaf derivednimbin is gaining traction due to the sustainability advantages associated with leaf harvesting, which does not require the destruction of the tree or the waiting period for seed maturation. According to the Journal of Ethnopharmacology, neem leaves contain trace amounts of nimbin along with other bioactive compounds such as azadirachtin and quercetin. This complex profile offers synergistic benefits that are attractive for certain cosmetic and herbal medicine applications. European producers are exploring leaf extraction as a way to diversify their supply sources and reduce reliance on seed imports. Leaf collection can be performed regularly throughout the year, providing a more consistent supply chain compared to seasonal seed availability. The extraction process for leaves often involves water or ethanol-based methods, which align with green chemistry principles. This appeals to brands marketing eco-friendly and ethically sourced ingredients. While the yield of pure nimbin from leaves is lower, advancements in extraction technology are improving efficiency.
REGIONAL ANALYSIS
Germany Nimbin Market Analysis
Germany was the largest contributor by holding 32.3% of the European market share in 2025, with its robust pharmaceutical industry and strong emphasis on phytotherapy. The country has a long-standing tradition of using herbal medicines, which is supported by a well-developed regulatory framework and extensive research infrastructure. This dominance translates into significant demand for high-quality botanical ingredients like nimbin for drug development. German consumers are highly educated about health and wellness, leading to high adoption rates of natural supplements and skincare products. The presence of major chemical and pharmaceutical companies facilitates the scaling up of nimbin extraction and formulation processes. Research institutions in Germany are actively investigating the clinical applications of nimbi,n particularly in dermatology and immunology. This scientific validation enhances consumer confidence and drives prescription trends. The country’s strict quality standards ensure that only premium-grade nimbin enters the market, which supports higher price points. Distribution channels are well organized, with pharmacies playing a central role in selling herbal remedies. The integration of Nimbin into statutory health insurance reimbursable products further boosts accessibility.
France Nimbin Market Analysis
France Nimbin market was next with 19.1% of the share in 2025. The growth of the market in France is mainly driven by the luxury and natural cosmetics sectors. The country is home to many of the world’s leading beauty brands, which are increasingly incorporating natural ingredients to meet consumer demand for clean and effective products. According to the French Federation of Beauty Companies, the export value of French cosmetics reached 16 billion euros in 2023, with natural products showing the highest growth rates. Nimbin is valued in French formulations for its soothing and anti-aging properties, which align with the brand image of elegance and efficacy. Consumer preferences in France lean towards premium products with transparent sourcing stories, which nimbin suppliers are leveraging. The popularity of spas and wellness centers in France also drives the use of nimbin in professional treatments. Marketing campaigns often highlight the exotic origin and traditional uses of neem to create an allure around the ingredient. The strong retail presence of parapharmacies provides a dedicated channel for selling nimbin-enriched skincare.
United Kingdom Nimbin Market Analysis
The United Kingdom's nimbin market growth is fueled by a vibrant nutraceutical sector and increasing health consciousness among its population. According to the Health Food Manufacturers’ Association, the UK vitamin and supplement market grew by 10% in 2023, reflecting a shift towards preventive healthcare. Nimbin is increasingly featured in immune-boosting supplements and anti-inflammatory capsules, catering to this trend. As per the Office for National Statistics, data e-commerce sales of health and beauty products increased by 15% year on year. This digital adoption allows smaller brands specializing in nimbin-based products to reach a wide audience without extensive physical retail networks. The presence of leading universities and research institutes supports ongoing studies into the health benefits of neem derivatives. These studies provide the scientific evidence needed to substantiate marketing claims and build consumer trust. The UK’s diverse population also contributes to a varied demand for traditional remedies, including those from Ayurvedic traditions, where neem is prominent. Retailers are expanding their natural health sections to accommodate this interest.
COMPETITION OVERVIEW
The competition in the European Nimbin market is characterized by a fragmented landscape with numerous small and medium-sized enterprises competing alongside established global suppliers. The market lacks dominant monopolies, which encourages innovation and competitive pricing among participants. Key competitors differentiate themselves through product quality, purity levels, and certification standards such as organic and non-GMO labels. The ability to provide consistent and standardized extracts is a critical factor in gaining trust from European pharmaceutical and cosmetic manufacturers. Companies are increasingly focusing on sustainability and ethical sourcing practices to align with the European Green Deal and consumer preferences. This shift has led to investments in transparent supply chains and eco-friendly extraction technologies. Regulatory compliance serves as a significant barrier to entry, ensuring that only reputable players with robust quality control systems can thrive. The market sees frequent collaborations between suppliers and research institutions to validate efficacy and explore new applications. Price sensitivity varies across segments, with pharmaceutical buyers prioritizing quality over cost, while agrochemical users seek cost-effective solutions. Innovation in formulation and delivery systems remains a key competitive edge.
KEY MARKET PLAYERS
A few major players of the European Nimbin market include
- EID Parry (India) Limited
- Neem Biotech
- Gramin India AgriBusiNest
- Ozone Biotech
- P.J. Margo Pvt. Ltd
- Fortune Biotech Ltd
- Agro Extracts Limited
- Nimtree Organics Pvt. Ltd
- The Indian Neem Tree Company
- Parker Biotech Private Limited
Top Strategies Used by the Key Market Participants
Key players in the Europe nimbin market primarily employ vertical integration strategies to secure raw material supplies and ensure consistent quality. Companies are increasingly investing in organic farming initiatives in source countries to guarantee sustainable and traceable neem seed procurement. This approach mitigates supply chain risks and appeals to environmentally conscious European consumers. Another major strategy involves technological advancement in extraction methods, such as supercritical fluid extraction, to produce higher purity nimbin without residual solvents. This enhances product appeal for premium pharmaceutical and cosmetic applications. Strategic partnerships and collaborations with local European distributors are also common, enabling faster market entry and improved customer support. Firms are actively engaging in clinical research to substantiate health claims, which builds trust with regulatory bodies and healthcare professionals. Additionally, companies are focusing on product differentiation by developing specialized nimbin formulations tailored to specific industry needs, such as anti-aging skincare or biopesticides.
Leading Players in the Europe Nimbin Market
- Margardit Oils Private Limited stands as a prominent supplier of neem-based extracts, including nimbin-rich fractions, to the European market. The company focuses on sustainable sourcing from certified organic farms in India, ensuring high purity and consistency in its product offerings. It has recently invested in advanced supercritical carbon dioxide extraction technologies to meet the stringent quality standards demanded by European pharmaceutical and cosmetic clients. This technological upgrade allows for solvent-free extracts, which align with the clean label trends prevalent in Europe. Margardit Oils actively collaborates with research institutions to validate the efficacy of its nimbin extracts through clinical studies. These efforts enhance credibility among healthcare professionals and formulators. The company also emphasizes traceability in its supply chain, providing transparency that is highly valued by European regulators and consumers.
- VedaOils has established a significant presence in the Europe nimbin market by offering a diverse range of neem-derived products tailored for personal care and wellness applications. The company leverages its extensive distribution network to ensure the timely delivery of high-quality nimbin extracts to manufacturers across Europe. Recent strategic initiatives include the expansion of its production facilities to increase capacity and improve efficiency. VedaOils has also launched specialized formulations that combine nimbin with other complementary botanicals to enhance therapeutic benefits. This innovation appeals to brands seeking unique selling propositions in a competitive landscape. The company actively participates in international trade fairs and industry conferences to showcase its capabilities and build relationships with potential clients. VedaOils prioritizes customer education by providing detailed technical data sheets and application guides.
- Indus Biotech is a key player in the Europe nimbin market, known for its expertise in the isolation and purification of bioactive compounds from neem. The company employs state of the art chromatography techniques to produce high-purity nimbin suitable for pharmaceutical and advanced cosmetic applications. Indus Biotech has recently strengthened its market position by entering into strategic partnerships with European distributors to expand its reach and improve customer service. These collaborations facilitate better market penetration and allow for more responsive supply chain management. The company invests heavily in research and development to explore new applications of nimbin in drug delivery systems and functional foods. Indus Biotech also adheres to rigorous quality control protocols, ensuring that every batch meets international standards.
MARKET SEGMENTATION
This research report on the Europe nimbin market has been segmented and sub-segmented based on application, source & region.
By Application
- Pharmaceutical & Nutraceutical Segment
- Cosmetics & Personal Care Segment
By Source
- Neem Seeds Segment
- Neem Leaves Segment
By Region
- UK
- France
- Spain
- Germany
- Italy
- Russia
- Sweden
- Denmark
- Switzerland
- Netherlands
- Turkey
- Czech Republic
- Rest of Europe
Frequently Asked Questions
1. What is the current growth trend of the Europe nimbin market?
The market is experiencing steady growth, driven by rising demand for plant-based ingredients and increasing adoption of herbal formulations across multiple industries.
2. What are the key drivers of the Europe nimbin market?
Major drivers include the growing preference for natural products, increased awareness of herbal medicine, expansion of clean beauty trends, and rising pharmaceutical research on plant-based compounds.
3. Which segment dominates the Europe nimbin market by application?
The pharmaceutical and nutraceutical segment dominates due to strong demand for natural therapeutic ingredients and immune-boosting supplements.
4. Which segment is growing the fastest in the Europe nimbin market?
The cosmetics and personal care segment is the fastest-growing, fueled by consumer demand for natural skincare and chemical-free formulations.
5. What are the primary sources of nimbin?
Nimbin is mainly extracted from neem seeds, which have the highest concentration, while neem leaves are emerging as a sustainable alternative source.
6. Why are neem seeds widely used for nimbin extraction?
Neem seeds offer higher yield, better purity, and consistent quality, making them the preferred source for industrial-scale extraction.
7. What challenges are faced by the Europe nimbin market?
Challenges include reliance on imported raw materials, fluctuations in supply quality, high extraction costs, and regulatory compliance requirements.
8. How is nimbin used in cosmetics and personal care products?
Nimbin is used in creams, serums, and cleansers for its antibacterial and anti-inflammatory properties, helping treat acne, irritation, and aging signs.
9. How is sustainability impacting the nimbin market?
Sustainability is encouraging the use of eco-friendly extraction methods, leaf-based sourcing, and ethical supply chains aligned with green chemistry principles.
10. What are the future opportunities in the Europe nimbin market?
Future opportunities include expansion in functional nutraceuticals, advanced drug formulations, sustainable beauty products, and emerging therapeutic applications.
Related Reports
Access the study in MULTIPLE FORMATS
Purchase options starting from
$ 2000
Didn’t find what you’re looking for?
TALK TO OUR ANALYST TEAM
Need something within your budget?
NO WORRIES! WE GOT YOU COVERED!
Call us on: +1 888 702 9696 (U.S Toll Free)
Write to us: sales@marketdataforecast.com